Group 1: Alkali Metals
The Party Animals of the Periodic Table ⚡
If there’s one group of elements that knows how to make a splash, it’s the alkali metals. Meet the wild bunch from Group 1: lithium, sodium, potassium, rubidium, caesium, and the mysterious (and dangerously radioactive) francium. These metals might seem chill sitting in the first column of the periodic table, but they’re anything but boring.
What Makes Them Special?
All Group 1 metals share a few key personality traits:
They only have one electron in their outermost shell.
That lone electron makes them super reactive—especially with water.
They’re soft—soft enough to slice with a butter knife.
Low melting points that get even lower as you go down the group.
The first three (lithium, sodium, potassium)? Less dense than water—so they float before they fizz!
When they do meet water, they form alkaline solutions (hence the name) and give off hydrogen gas.
Because they love reacting with oxygen and water, alkali metals are stored in oil, keeping them from throwing spontaneous science experiments in the lab.
Reactivity: Why the Drama Increases Down the Group
You’d think the bigger the atom, the lazier it’d get—but nope! In Group 1, the reactivity increases as you go down:
Each new element adds another electron shell, so the outer electron gets further from the nucleus.
That means it feels less pull from the positively charged nucleus. So it takes less energy to kick that outer electron out of the party. Result? It’s easier to lose that electron and form a 1+ ion.
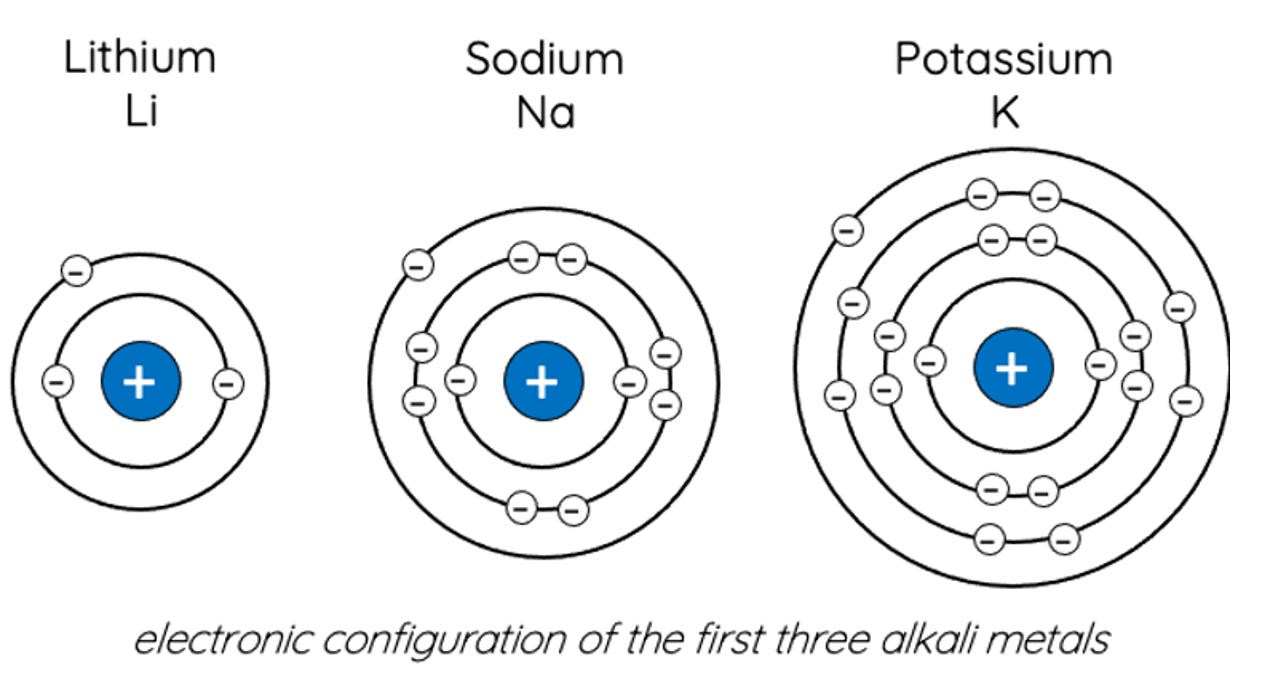
Here’s the breakdown using electron configurations (the leftmost number refers to the electron shell nearest the nucleus):
Lithium (Li) – 2,1
Sodium (Na) – 2,8,1
Potassium (K) – 2,8,8,1
…and so on, getting larger and looser with every step down
Water Reactions: The Showstoppers
Drop an alkali metal into water and prepare for a science show:
Lithium: fizzes gently
Sodium: zips around the surface
Potassium: sparks and lilac flames
Rubidium & Caesium: EXPLOSIVE
They all form metal hydroxides, making the water alkaline, and release hydrogen gas.
... and francium?
Francium is the rare beast of the group. So unstable, so radioactive, and so elusive that you’re more likely to find a unicorn riding a rainbow than see a chunk of francium reacting with water.
Next time you’re scanning the periodic table, give a nod to Group 1—the metals that cut like butter, float like corks, and react like rockstars. (Just… maybe like a mogwai, don’t take them to a pool party!).